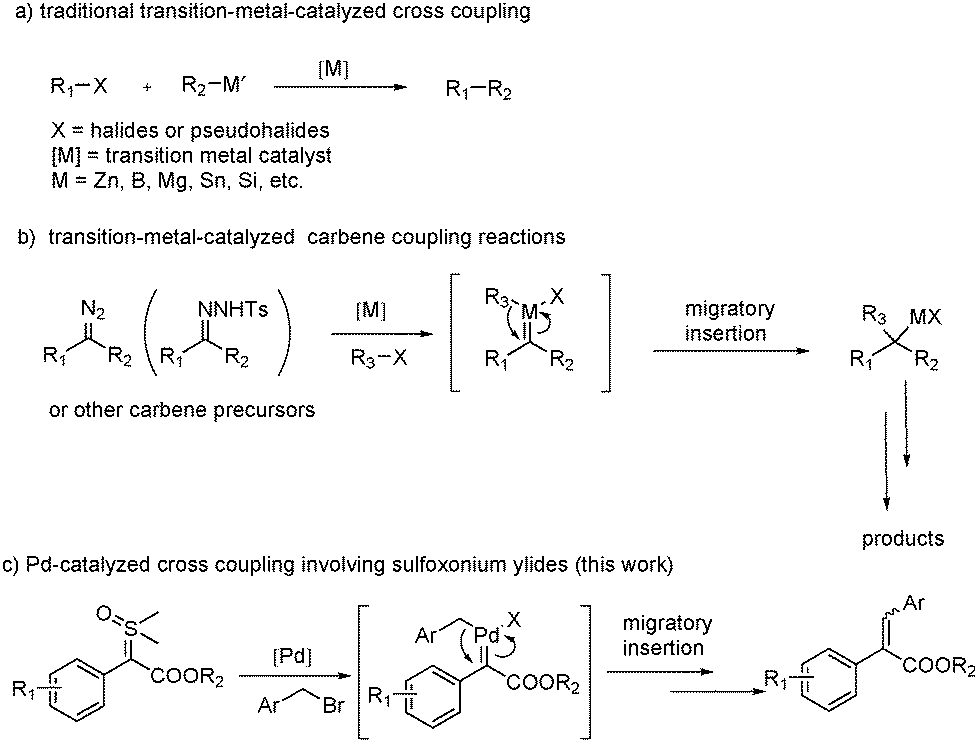
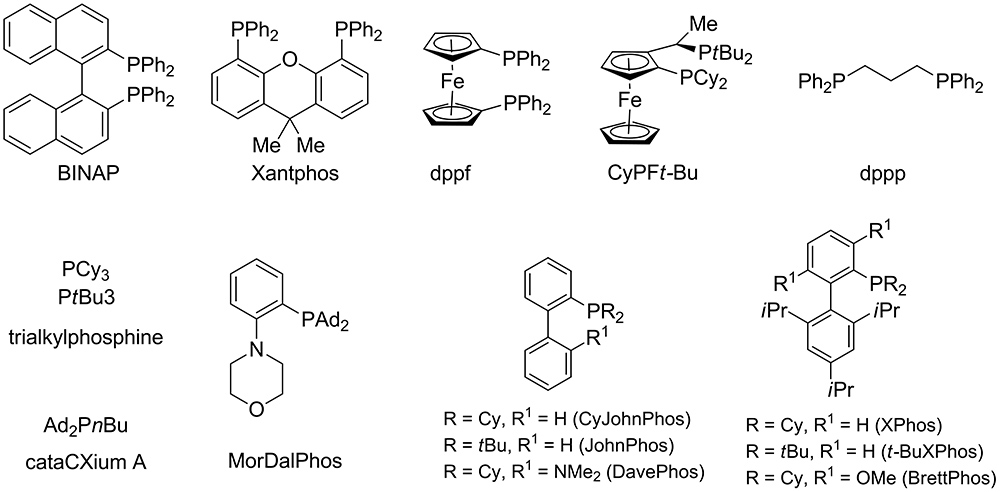
The role of allyl ammonium salts in palladium-catalyzed cascade reactions towards the synthesis of spiro-fused heterocycles | Nature Communications

Full article: Palladium-catalyzed coupling reactions in flavonoids: A retrospective of recent synthetic approaches
![Catalysts | Free Full-Text | Palladium-Catalyzed Three-Component Coupling of Benzynes, Benzylic/Allylic Bromides and 1,1-Bis[(pinacolato)boryl]methane Catalysts | Free Full-Text | Palladium-Catalyzed Three-Component Coupling of Benzynes, Benzylic/Allylic Bromides and 1,1-Bis[(pinacolato)boryl]methane](https://www.mdpi.com/catalysts/catalysts-13-00126/article_deploy/html/images/catalysts-13-00126-sch001.png)
Catalysts | Free Full-Text | Palladium-Catalyzed Three-Component Coupling of Benzynes, Benzylic/Allylic Bromides and 1,1-Bis[(pinacolato)boryl]methane

Palladium-Catalyzed Cross-Coupling Reactions of Organosilanols and Their Salts: Practical Alternatives to Boron- and Tin-Based Methods | Accounts of Chemical Research

Synthesis of Allyl Cyanamides and N-Cyanoindoles via the Palladium-Catalyzed Three-Component Coupling Reaction | Journal of the American Chemical Society

A General and Efficient Catalyst for Palladium-Catalyzed C−O Coupling Reactions of Aryl Halides with Primary Alcohols | Journal of the American Chemical Society

Palladium-Catalyzed C–P Bond-Forming Reactions of Aryl Nonaflates Accelerated by Iodide - ScienceDirect
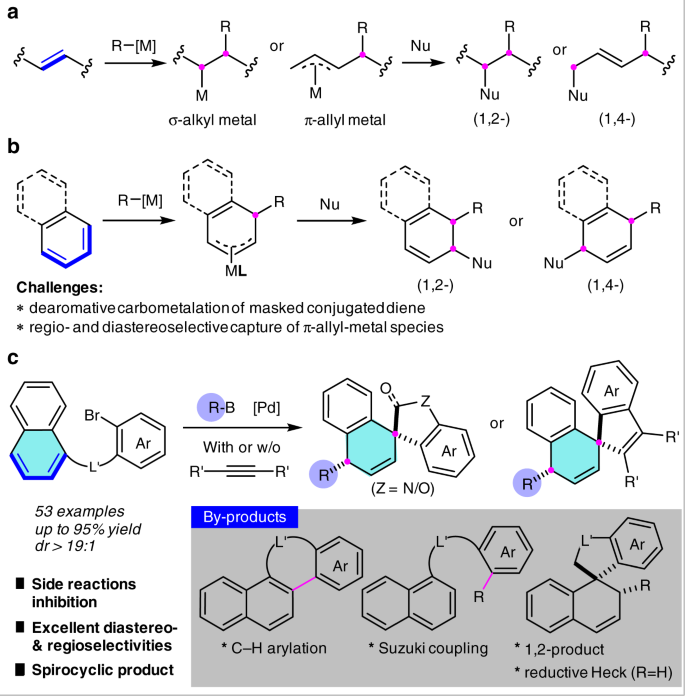
Dearomative 1,4-difunctionalization of naphthalenes via palladium-catalyzed tandem Heck/Suzuki coupling reaction | Nature Communications

Palladium-Catalyzed Allylic Carbonylative Negishi Cross-Coupling Reactions with Sterically Bulky Aromatic Isocyanides